This page was produced as an assignment for Genetics 677, a undergraduate course at The University of Wisconsin - Madison
CAPN3-Protein Interactions and Pathways
What does the protein-interaction network of CAPN3 look like?
________________________________________________________________________________________________________
CAPN3 is a skeletal muscle-specific, weak cysteine protease that is known to interact with Titin in the sarcomere. Its mild proteolytic ability suggests that its role is protein-modification rather than degradation, and its interaction with Titin is crucial to sarcomere turnover and remodeling, and the elasticity of the sarcomere [2,3,4].
Since the initial linking of CAPN3 to LGMD2A, many more of its subtrates and interaction partners have been, and continue to be, discovered. To find out more about the currently known and predicted CAPN3 interactome, I first looked up the entry for human calpain-3 protein (CAN3_HUMAN) on STRING).
CAPN3 Human Interactome (from STRING)
Legend for CAPN3 Human Interactome (from STRING)
The STRING map shows experimental evidence for CAPN3 interaction with essential sarcomeric proteins (Titin/TTN, Dysferlin/DYSF), the small, regulatory subunit of uqiquitous calpains (CAPNS1), and is predicted to interact with Dystrophin (DMD), the gene associated with Duchenne Muscular Dystrophy.
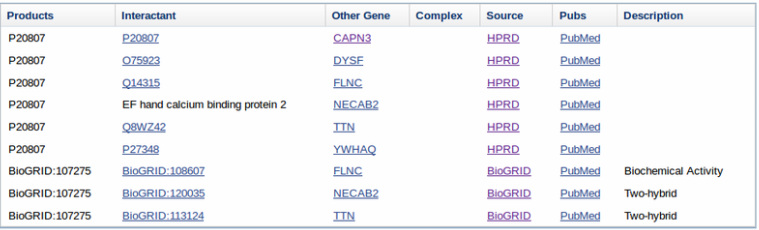
A list of interacting proteins was also found on the GenBank page for human CAPN3 (below) and I compared them to my results from STRING.
Proteins Interacting with CAPN3 (from GenBank)
Discussion
These results provide strong evidence for the central role of CAPN3 within the sarcomere and its function in the regulation of sarcomere-remodeling and turnover. Interaction with Titin and Dysferlin is shown in both STRING and on GenBank, but the other proteins listed on each site are not consistent with one another.
The importance of CAPN3-TTN interaction, in which CAPN3 is believed to proteolytically regulate TTN, has been estabished as a key step to activation of TTN and its role in regulating muscle elasticity and renewal [4].
Recent work has tied CAPN3 to a central role in the cell fate-governing NFκ-B pathway as a regulator of IkB regulatory factors (Figure 3) [5,7]. Furthermore, Muscle Ankyrin Repeat Proteins (MARPs), such as CARP (Cardiac ARP) and Ankrd2 have more recently been established as CAPN3 substrates (see below) [5].
The importance of CAPN3-TTN interaction, in which CAPN3 is believed to proteolytically regulate TTN, has been estabished as a key step to activation of TTN and its role in regulating muscle elasticity and renewal [4].
Recent work has tied CAPN3 to a central role in the cell fate-governing NFκ-B pathway as a regulator of IkB regulatory factors (Figure 3) [5,7]. Furthermore, Muscle Ankyrin Repeat Proteins (MARPs), such as CARP (Cardiac ARP) and Ankrd2 have more recently been established as CAPN3 substrates (see below) [5].
The NF-kB Signaling Pathway in Muscle Cells [6]

ABOVE: This diagram shows Calpain-3 in the NF-kB cell-fate determining signaling cascade of muscle cells. Normally, CAPN3 would be activated by calcium ions to degrade IkB, releasing NF-kB p50 and p65 from sequestration and thus allowing them to enter the nucleus and regulate survival genes. When CAPN3 is absent, as in LGMD2A, gene regulation by p50/p65 is thrown off, manifesting in deregulation of NF-kB-mediated apoptosis and survival.
________________________________________________________________________________________________________
|
References:
[1] Chung, CS, Morton, NE. Discrimination of genetic entities in muscular dystrophy. Am. J. Hum. Genet. 11: 339-359, 1959. [2] Fanin, M et al. Molecular diagnosis in LGMD2A: mutation analysis or protein testing? Hum. Mutat. 24: 52-62, 2004. [3] Jensen LJ, et al. STRING 8--a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res. 2009 Jan;37 [4] Kinbara K, Sorimachi H, Ishiura S, Suzuki K. Muscle-specific calpain, p94, interacts with the extreme C-terminal region of connectin, a unique region flanked by two immunoglobulin C2 motifs. Arch Biochem Biophys. 1997 Jun 1;342(1):99-107 [5] Laure, L. et al. A new pathway encompassing calpain 3 and its newly identified substrate cardiac ankyrin repeat protein is involved in the regulation of the nuclear factor-κB pathway in skeletal muscle. FEBS J. 277, 4322–4337 (2010). |
[6] Mourkioti, F. & Rosenthal, N. NF-κB signaling in skeletal muscle: prospects for intervention in muscle diseases. Journal of Molecular Medicine 86, 747–759 (2008). [7] Miller MK, Bang ML, et al. (2003). The Muscle Ankyrin Repeat Domain Proteins: CARP, anrkd2/Arpp, and DARP as a family of Titin Filament-based Stress Response Molecules. J. Mol. Biol. 333, 951-964. [8] Richard, I. et al. Loss of calpain 3 proteolytic activity leads to muscular dystrophy and to apoptosis-associated IkappaBalpha/nuclear factor kappaB pathway perturbation in mice. J. Cell Biol. 151, 1583–1590 (2000). |